The Definition of Exploratory Journals
It is believed that sharing exploratory and novel research results, as well as innovative and enlightened ideas or hypotheses on specific topics in the form of official publication is very important, as this not only incites more extensive and fruitful research on these topics, but also engraves a historical hallmark in the journal. To this end, XHP established two journals focusing on exploratory research and hypothesis, namely Exploratory Journals, including Exploratory Research and Hypothesis in Medicine, and Journal of Exploratory Research in Pharmacology.
The Unique Character of Manuscripts Published in Exploratory Journals
Apart from the standard sections for original articles (Introduction, Methods and Materials, Results, and Discussion [IMRD]) or review articles, an additional section of “Future Research Directions/ Prospect/ Prediction”, with or without “Hypothesis”, with no less than 100 words, is REQUIRED by the Exploratory Journals. However, although not required, a “Hypothesis” paragraph is highly preferred. This section is preferably accompanied by a graph that illustrates the exploratory research or hypothesis (e.g., proposed molecular mechanisms).
The proposed hypothesis or future research directions/prospect/prediction should be derived from the results of the study, although it may be preliminary, incomprehensive, or incomplete, with direct or indirect support from previous research findings in the literature. Thus, this section should shed new light on further basic and translational research, and may provide clues to approaches to clinical application. Some examples are provided below.
Examples 1:
Future research predictions
The use of genotypic information in personalized nutrition offers considerable future promise, but significant barriers exist to successful implementation, independent of scientific knowledge. These include consumer acceptance, ethical, technological, and regulatory considerations. Research into nutrigenetics has produced inconsistent results; however, the same could be said for conventional nutrition studies. This is not necessarily due to the overall quality of the research and the magnitude of the body of knowledge, rather it is due to the complex interactions between nutrition, genetics, and long-term health. Improved frameworks are required to translate nutrigenetic studies into usable guidelines to direct practicing nutrition and medical professionals. This will require an interdisciplinary approach, including geneticists, bioinformaticians, nutritionists, dietitians and other biomedical professionals.
Furthermore, additional research is needed not only into the gene-nutrient interactions themselves but also into the public attitudes and acceptance of nutrigenetics and the associated risks and benefits of uptake. Without a holistic approach to implementation, it is unlikely that nutrigenetics will deliver on its early promise to improve health outcomes.
Examples 2:
Future research directions
It is hypothesized that consumption of FA may result in decreased neurotransmitter production due to decreased availability of biologically active folate 5-MTHF and a resulting decrease in SAMe. Moreover, the addition of FA into a finely balanced yet complicated cycle in susceptible individuals (e.g., those with MTHFR, MTR, MTRR and COMT polymorphisms) may serve to greatly slow the cycle via the inhibition of MTHFR by DHF. As the population continues to consume less folate from food sources such as leafy green vegetables, then it is postulated that the rates and severity of depression will increase as 5-MTHF decreases. Abbreviations: DHF, dihydrofolate; FA, folic acid; 5-MTHF, 5-methyltetrahydrofolate; MTHFR, methylenetetrahydrofolate reductase; SAMe, S-adenosylmethionine.
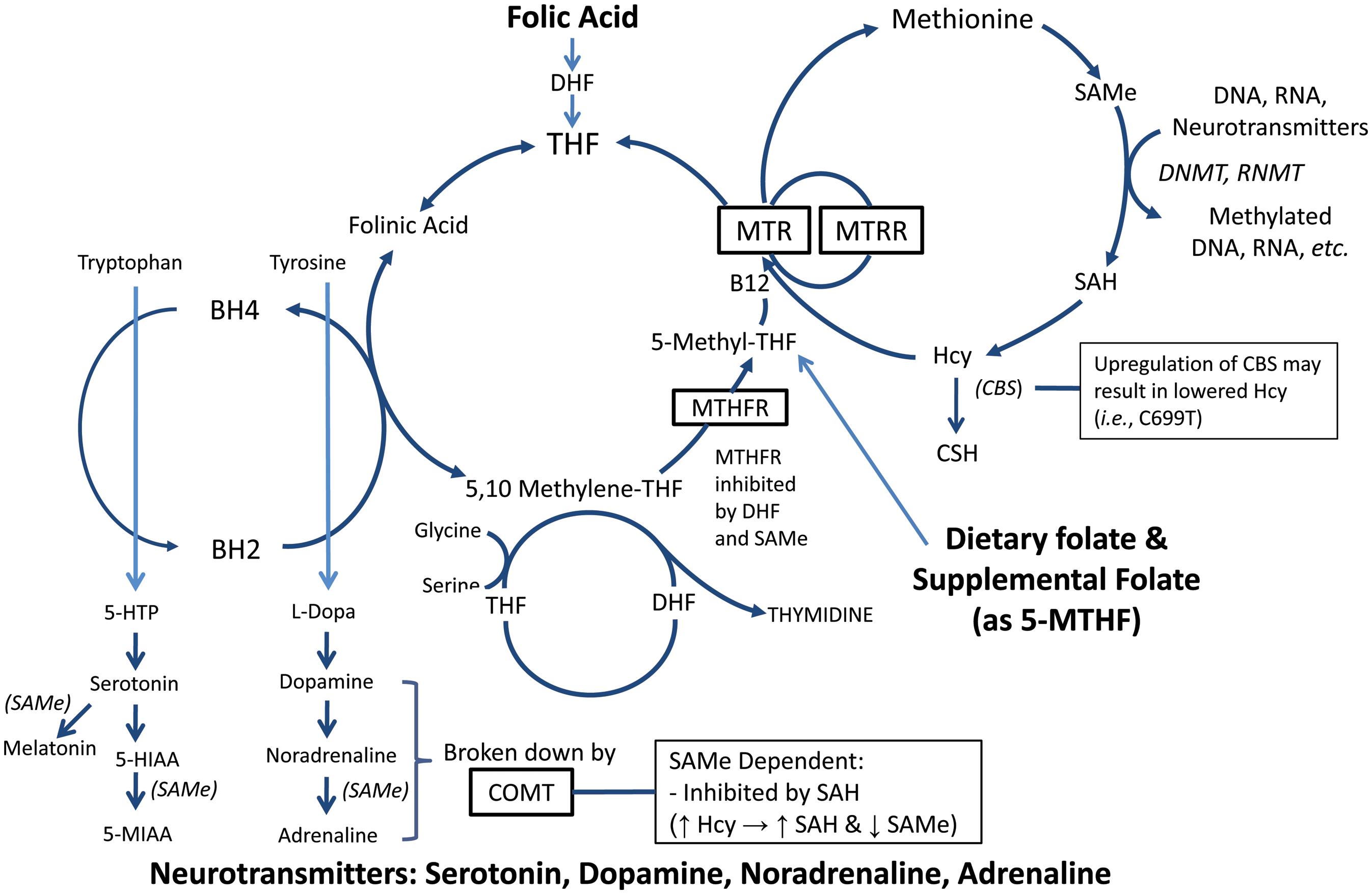
Fig. 2 Relationship between folate, methylation and neurotransmitters involved with depression.
Examples 3:
Future research directions
Further research is necessary to either confirm or refute the presently observed associations of VDR gene variants with cognitive decline as measured using the MMSE scale. By obtaining a more thorough understanding of VDR variants and their influence on risk of cognitive decline, new insights into the underlying pathophysiology of cognitive decline and development of possible intervention and treatment strategies will emerge. These may include the screening of particular VDR polymorphisms as part of a routine health check and the use of supplemental vitamin D at a younger age. Based on the preliminary results of this study, we hypothesize that the use of vitamin D as a potential preventative agent in cognitive decline will reduce the impact this degenerative disorder currently has on our health system.
Examples 4:
Hypothesis
By increasing the amounts of serotonin in bone marrow, SSRIs may improve the efficacy of HSC transplantation (Fig. 1). Most studies concerning SSRIs have focused on the effects of sertraline and fluoxetine. Because the effects of fluoxetine on lymphocytes are variable under different conditions, we suggest sertraline, as its effect is solely immunosuppressive. The routine starting dosage of sertraline as an antidepressant is 50 mg/day, and the maximum is 200 mg/day,29,30 but higher doses are probably required for immunosuppression.19 The normal concentration of serotonin in plasma is 0.62±0.11 μg/L.31 Future studies must focus on the dosage of sertraline that will generate the optimum concentration of plasma serotonin for in vivo expansion of HSCs.
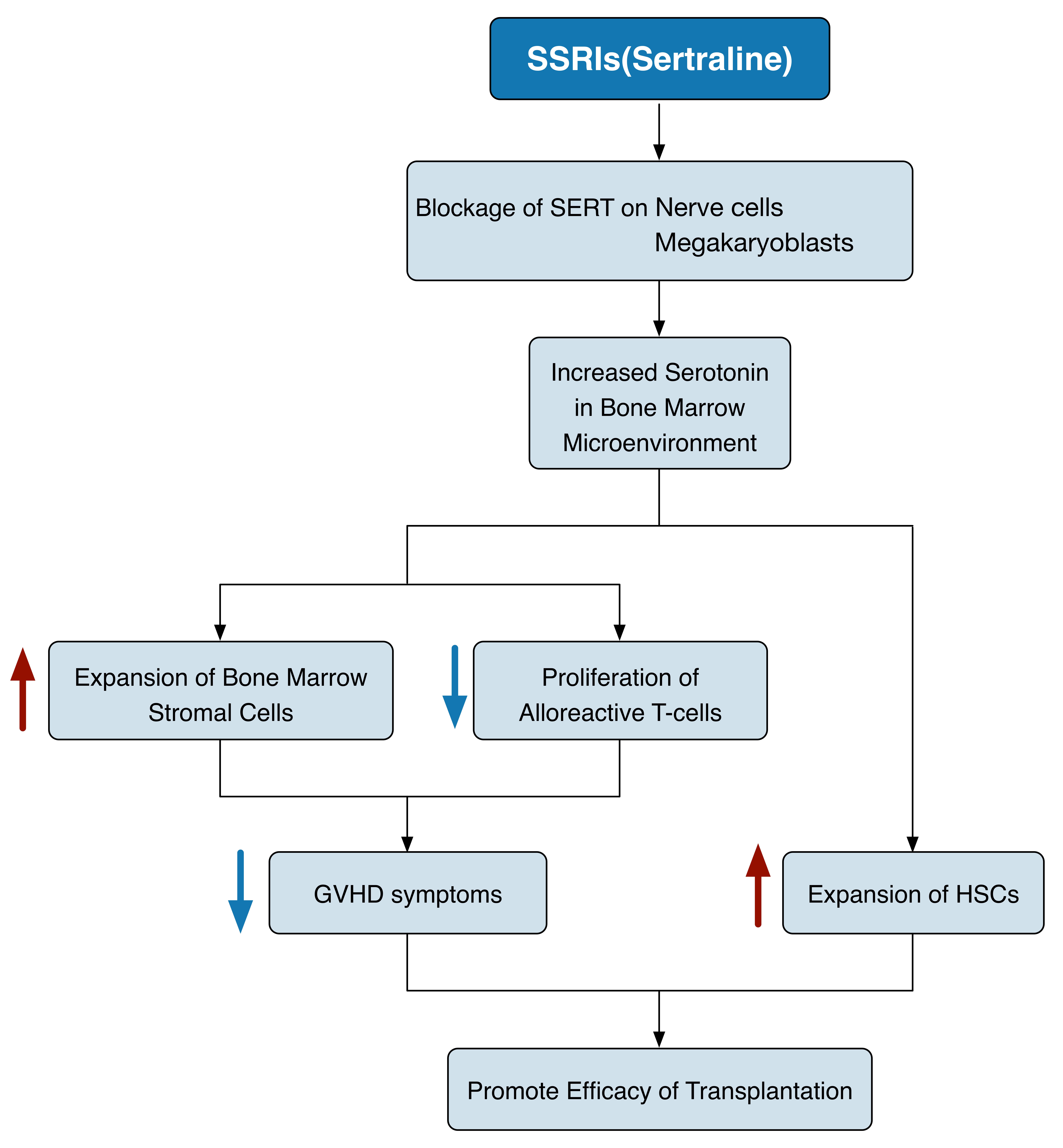
Fig. 1 Schematic diagram of the hypothetical mechanisms underlying SSRI promotion of HSC transplantation through in vivo expansion of HSCs and decrease in GVHD.
Controversial or Negative Results
JERP also publishes original articles with “non-confirmatory” or “negative” data that are generated from appropriate methods and materials, and refute a proposed hypothesis or challenge a currently accepted hypothesis. Systematic review and meta-analysis reporting “non-confirmatory” or “negative” findings are acceptable.
Examples of negative results
1. Takanashi S, Saif LJ, Hughes JH, Meulia T, Jung K, Scheuer KA, Wang Q. Failure of propagation of human norovirus in intestinal epithelial cells with microvilli grown in three-dimensional cultures. Arch Virol. 2014 Feb;159(2):257-66. doi: 10.1007/s00705-013-1806-4
2. Svanstrom H, Pasternak B, Hviid A. Use of azithromycin and death from cardiovascular causes. N Engl J Med. 2013 May 2;368(18):1704-12. doi: 10.1056/NEJMoa1300799
3. Whitehead KJ, Sautter NB, McWilliams JP, Chakinala MM, Merlo CA, Johnson MH, James M, Everett EM, Clancy MS, Faughnan ME, Oh SP,Olitsky SE, Pyeritz RE, Gossage JR. Effect of Topical Intranasal Therapy on Epistaxis Frequency in Patients with Hereditary Hemorrhagic Telangiectasia: A Randomized Clinical Trial. JAMA. 2016 Sep 6;316(9):943-51. doi:10.1001/jama.2016.11724
Example of controversial research:
Xia HH, Talley NJ, Kam EP, Young LJ, Hammer J, Horowitz M. Helicobacter pylori infection is not associated with diabetes mellitus, nor with upper gastrointestinal symptoms in diabetes mellitus. Am J Gastroenterol. 2001 Apr;96(4):1039-46. doi: 10.1111/j.1572-0241.2001.03604.x
Examples of refuting/challenging hypotheses:
1. Abbas AK. Protein Synthesis Inhibitors Did Not Interfere with Long-Term Depression Induced either Electrically in Juvenile Rats or Chemically in Middle-Aged Rats. PLoS One. 2016 Aug 12;11(8):e0161270. doi: 10.1371/journal.pone.0161270
2. Xia HH, Talley NJ, Kam EP, Young LJ, Hammer J, Horowitz M. Helicobacter pylori infection is not associated with diabetes mellitus, nor with upper gastrointestinal symptoms in diabetes mellitus. Am J Gastroenterol. 2001 Apr;96(4):1039-46. doi: 10.1111/j.1572-0241.2001.03604.x
3. Gunnarsson H, Arason A, Gillanders EM, Agnarsson BA, Johannesdottir G, Johannsson OT, Barkardottir RB. Evidence against PALB2 involvement in Icelandic breast cancer susceptibility. J Negat Results Biomed. 2008 Jul 17;7:5. doi: 10.1186/1477-5751-7-5
