Cytopathology Reporting Systems provide standardized language for reporting in cytopathology practice. Standardized reporting categories allow effective and consistent communications across different institutions and disciplines, and convey unambiguous and clinically helpful information to our clinicians. Since the establishment of the Bethesda System for Reporting Cervical Cytology, other systems have followed and proved successful. The current updated Cytopathology Reporting Systems include: 1. The Bethesda System for Reporting Cervical Cytology (3rd ed, 2015)
2. The Bethesda System for Reporting Thyroid Cytopathology (3rd ed, 2023)
3. The Milan System for Reporting Salivary Gland Cytopathology (2nd ed, 2023)
4. The Paris System for Reporting Urinary Cytology (2nd, 2022)
5. The International Academy of Cytology Yokohama System for Reporting Breast Fine Needle Aspiration Biopsy Cytopathology (1st ed, 2020)
6. The International System for Reporting Serous Fluid Cytopathology (1st ed, 2020)
7. The WHO Reporting System for Lung Cytopathology (1st ed, 2023)
8. The WHO Reporting System for Pancreaticobiliary Cytopathology (1st ed, 2023)
9. The WHO Reporting System for Lymph Node Cytopathology
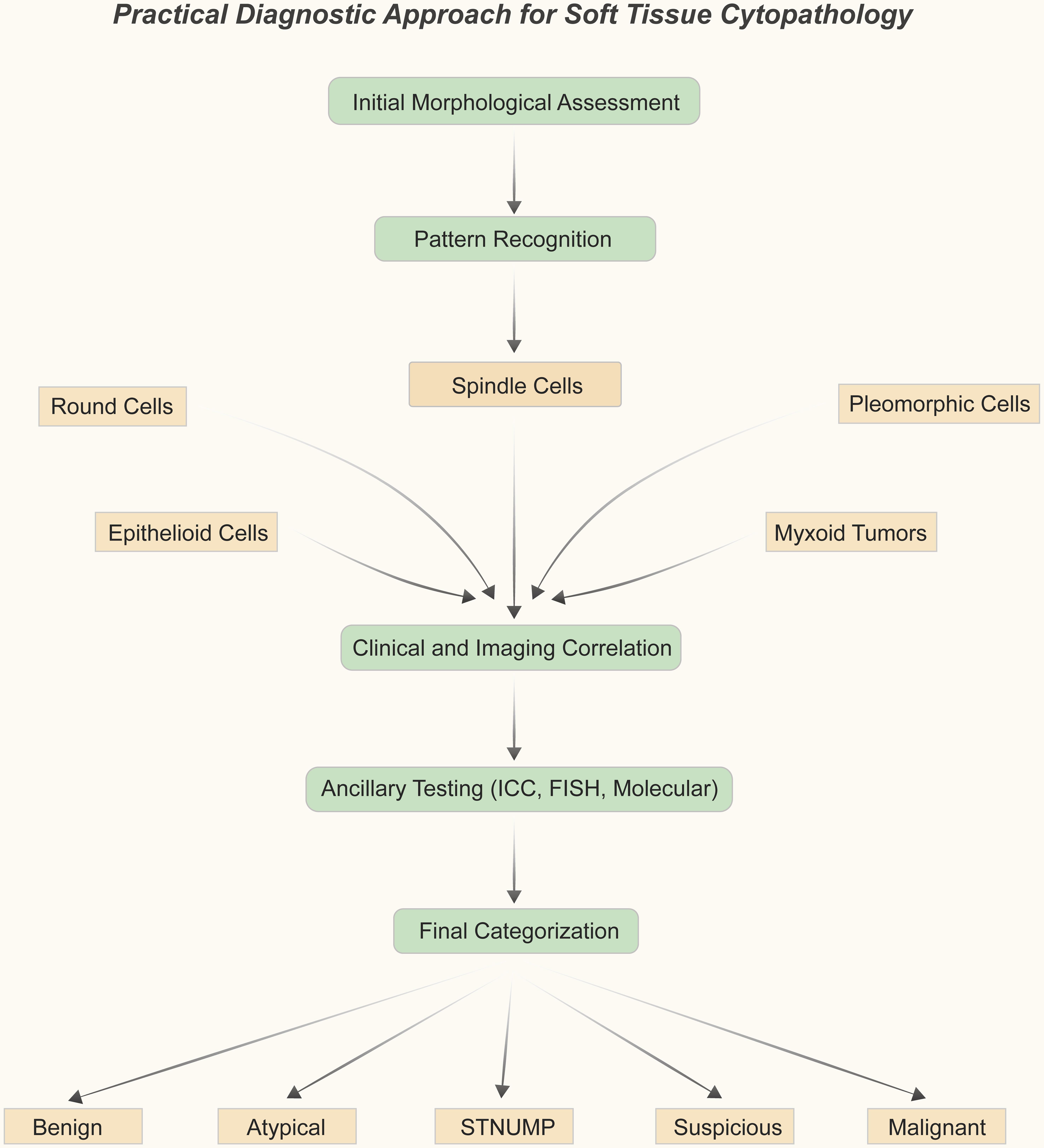
10. The WHO Reporting System for Soft Tissue Cytopathology
Review Article
|
World Health Organization Reporting System for Soft Tissue Cytopathology: A Concise Review with a Practical Diagnostic Approach
Marilyn M. Bui
Published online: June 24, 2025
doi:10.14218/JCTP.2025.00016
|
|
The World Health Organization System for Reporting Pancreaticobiliary Cytopathology: Standardized Categories and Practical Approaches to Pancreatic Lesions
Minhua Wang, Maria D. Lozano, Guoping Cai
Published online: September 25, 2024
doi:10.14218/JCTP.2024.00034
|
|
The World Health Organization Reporting System for Lung Cytopathology–A Review of the First Edition
Darin Dolezal, Ivana Kholová, Guoping Cai
Published online: March 11, 2024
doi:10.14218/JCTP.2023.00068
|
|
The International System for Reporting Serous Fluid Cytopathology—An Updated Review
Minhua Wang, Ashish Chandra, Guoping Cai
Published online: December 20, 2023
doi:10.14218/JCTP.2023.00025
|
Mini Review
|
Introduction of the WHO Reporting System for Lymph Node Cytopathology
Yan Gao, Sara E. Monaco, Ruth L. Katz, Y. Helen Zhang
Published online: December 23, 2023
doi:10.14218/JCTP.2023.00044
|
Editorial
![]()
|
The Evolution of Cytopathology Reporting: Standardization, Precision, and Future Directions
Fernando Schmitt
Published online: June 24, 2025
doi:10.14218/JCTP.2025.00015
|
|
|

|
Guest Editor
Fang Fan
|
Read More
|
|
Dr. Fang Fan earned her medical degree from Shanghai Medical School at Fudan University in China. She then began her two decades-long association with the University of Kansas Medical Center in Kansas City, Kansas, earning a Ph.D., completing her pathology residency and cytopathology fellowship, staying as a faculty, and ultimately rising to Director of Cytopathology and Barbara F. Atkinson Endowed Professor at the Pathology department. Dr. Fan joined City of Hope pathology department in 2021 and she is currently Clinical Professor and Director of Cytopathology at City of Hope Medical Center in Duarte, California.
Dr. Fan has more than 100 scholarly articles and many national presentations in the fields of cytopathology and breast pathology. She has given Cytopathology lectures/Microscopic sessions in the Oakstone Lecture Series, Osler Cytopathology Board Review courses, CAP annual meetings, ASC annual meetings, and USCAP annual meetings. She is the chief editor of a Cytopathology Review book which is currently in its third edition. Dr. Fan has served on the Cytopathology Committee of the College of American Pathologists (CAP), and Membership Committee, International Liaison Committee, and Scientific Program Committee of the American Society of Cytopathology (ASC).
|

|
Guest Editor
Guoping Cai
|
Read More
|
|
Dr. Cai earned his medical degree from Wenzhou Medical Univeristy in China. He completed his residency in Anatomic and Clinical Pathology at New York University Medical Center, where he also completed a fellowship in cytopathology. He is a board-certified pathologist and cytopathologist. He is currently Professor of Pathology and Associate Director of Cytopathology in the Department of Pathology, Yale University School of Medicine.
Dr. Cai's research interest includes molecular and biomarker testing in lung, pancreas, and thyroid cancers. He is a member of the Cancer Signaling Networks Program at Yale Cancer Center. He has published over 120 peer-reviewed articles. He is an editor for the book entitled “Rapid On-site Evaluation: A Parctical Guide”, and is a contributor for the WHO Reporting System for Pancreaticobiliary Cytopathology. He currently serves on the Progressive Evaluation Committee of American Society of Cytopathology (ASC) and the Membership Committee of International Association for the Study of Lung Cancer (IASLC).
|
Important Dates
Submission open date: May 1, 2023
Submission deadline: November 30, 2023
|